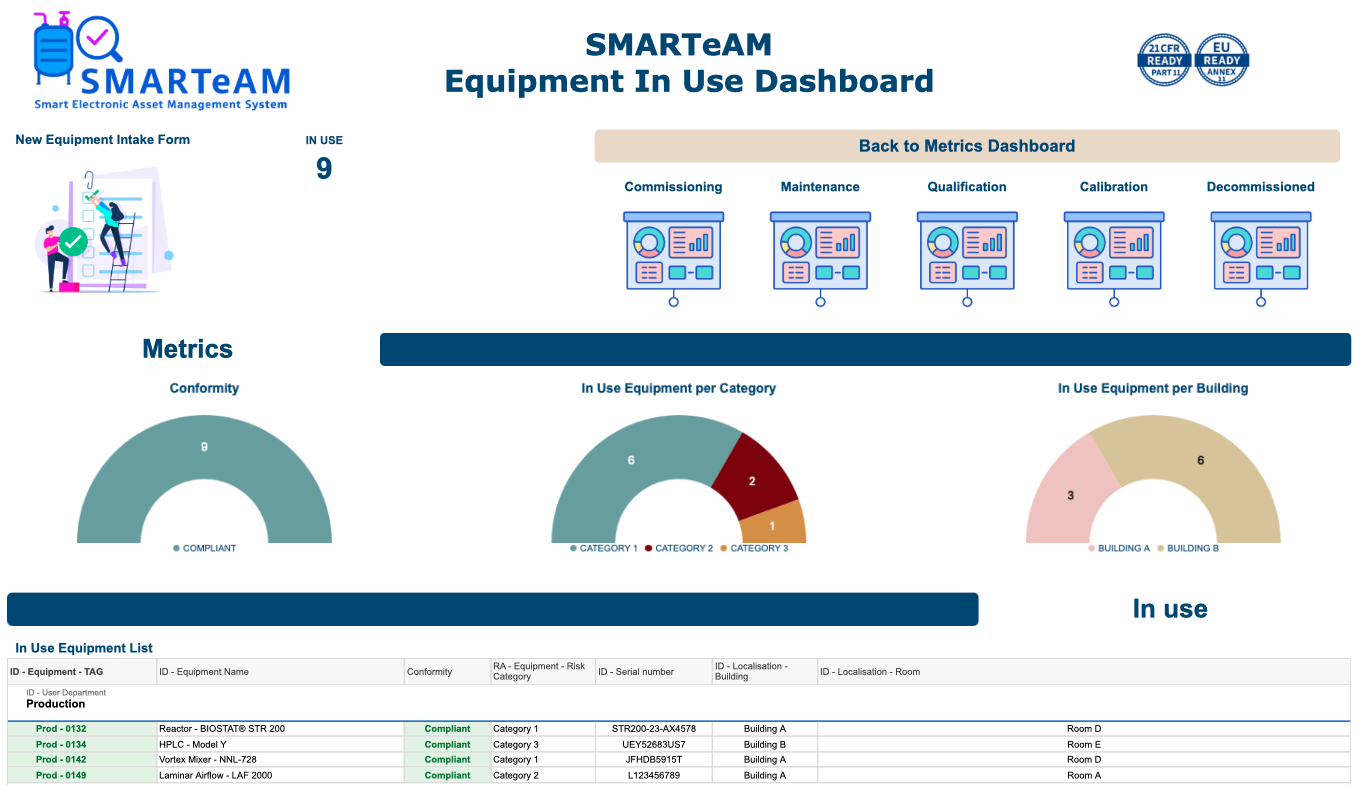
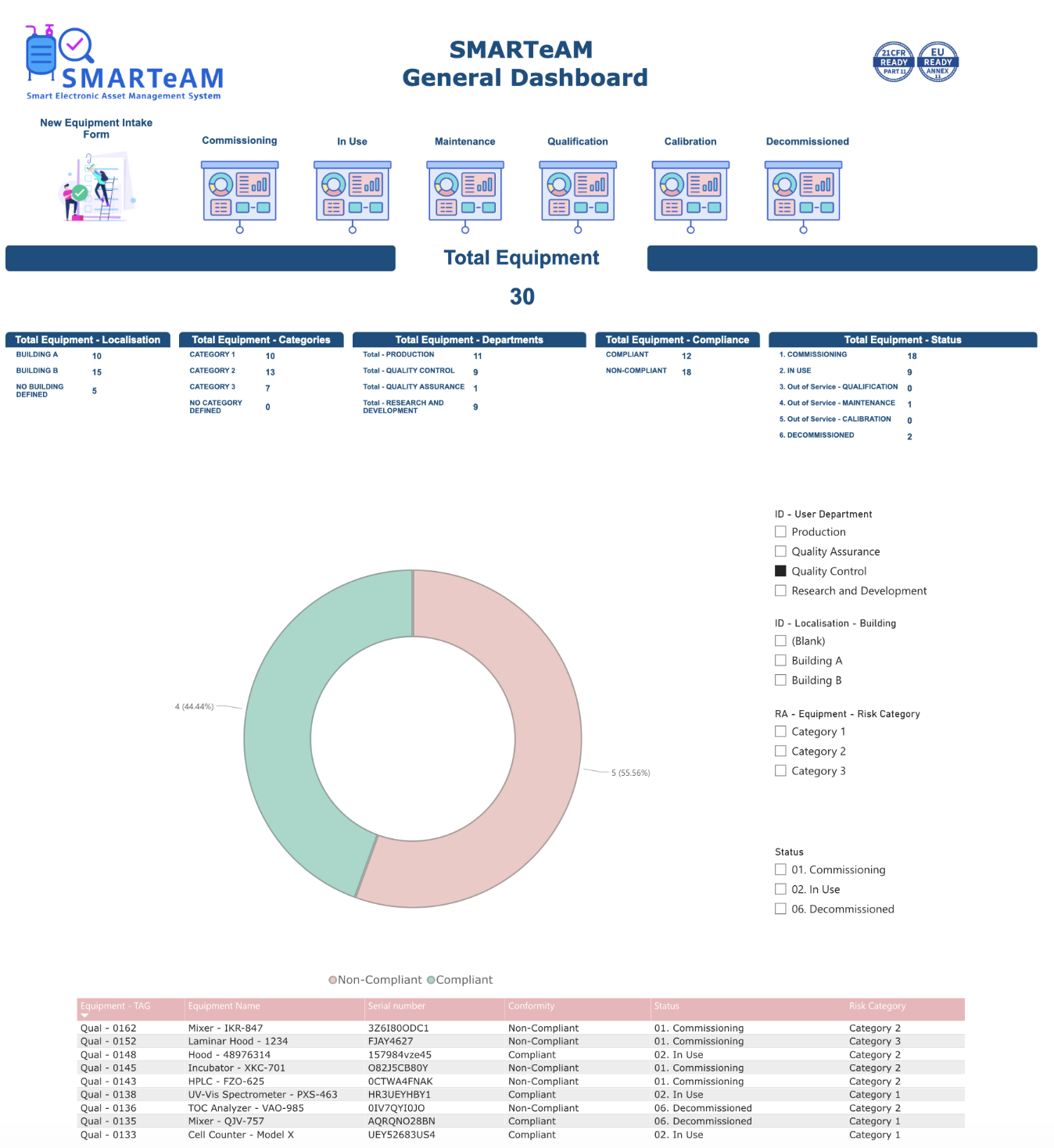
This dashboard provides a centralized view of your equipment fleet, offering key metrics on compliance, status, and departmental distribution. It serves as a hub to navigate to more detailed dashboards for specific equipment conditions. Integrated with Power BI, the dashboard delivers interactive charts and real-time insights, enabling users to filter data by compliance, status, or department. A dropdown list allows quick access to detailed information for individual equipment. By leveraging Smartsheet’s connectors, this solution integrates seamlessly with other tools and systems, automates workflows, and ensures data accuracy without duplication. The dashboard helps teams maintain compliance, prevent risks of malfunction, and reduce downtime—keeping your equipment fleet running efficiently.